Journal reference: Physical Review X, DOI: 10.1103/PhysRevX.11. 
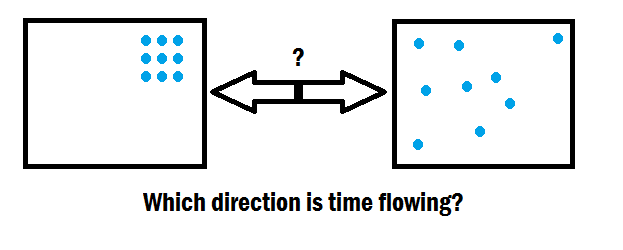
More specifically, the second law of thermodynamics states that as one goes forward in time, the net entropy (degree of disorder) of any isolated or closed system will always increase (or at least. This finding could also help us understand more generally how the quantum world and the classical world are similar and different in terms of thermodynamics and the passage of time. Entropy, a measure of disorder, explains why life seems to get more, not less, complicated as time goes on. Understanding this relationship could be helpful for designing clocks in the future, particularly those used in quantum computers and other devices where both accuracy and temperature are crucial, says Ares. “It’s still unclear how this principle plays out in real devices such as atomic clocks, which push the ultimate quantum limits of accuracy,” says Mark Mitchison at Trinity College Dublin in Ireland. 
We can’t be sure yet that these results are actually universal, though, because there are many types of clocks for which the relationship between accuracy and entropy haven’t been tested. “I think it’s hinting at this universality of how the laws of thermodynamics apply to clocks,” says Ares. When the researchers compared their results with theoretical models developed for clocks that rely on quantum effects, they were surprised to find that the relationship between accuracy and entropy seemed to be the same for both. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
